
Social Media for Healthcare
Social media for healthcare: How to keep patients engaged in the digital age
Navigating the complexities of social media in healthcare requires a delicate balance of building reputation, raising public health awareness and ensuring HIPAA compliance. Learn more about how to leverage its potential for community engagement and talent acquisition, and how Sprout can help you do that at scale.
Reading time 9 minutes
Published on October 27, 2025

Table of Contents
Summary
- Social media is a versatile tool for healthcare, serving as a patient acquisition, brand management and public awareness platform.
- Despite its benefits, leveraging social media effectively in healthcare is challenging due to industry regulations and unique objectives.
- Successful use of social media in healthcare can enhance reputation, raise public health awareness, mitigate crises, attract talent and improve communication.
The applications of social media for healthcare are endless. It’s a new patient acquisition tool, a brand management tool and a public awareness tool all rolled into one.
Nevertheless, harnessing its power is notoriously challenging. The healthcare industry is highly regulated, with distinct target audiences and specific objectives. Most social media marketing advice primarily focuses on industries that offer clearer paths to return on investment (ROI), leaving healthcare marketers on their own when building strategies.
When healthcare organizations succeed on social media, they help communities succeed in the real world. We put together this guide to support those missions and supply you with the best practices and advice needed to grow your audience on social.
What are the benefits of social media in healthcare?
Social media in healthcare delivers five critical benefits: building community trust and reputation, educating patients on public health topics, managing crisis communications, attracting top medical talent and gathering patient insights through social listening. These capabilities transform how healthcare organizations connect with patients and communities.
Build your reputation within a community
Social can play a significant role in helping healthcare organizations effectively reach diverse audiences across generations. Through social, healthcare providers can engage in direct, transparent communication with a community, whether they be local, circumstantial or interest-based.
This active engagement acts as a form of reputation management, positioning your organization as a reliable provider of healthcare services and information. This way, when a patient is exploring care options, your organization is top of mind.
The concept of competition in the health sector is a touchy one. Even so, any given speciality is full of providers and it’s only natural for prospective patients to “shop around.”
It only makes sense for healthcare marketers to present their practices as leaders in their respective specialties. From showing off employees in action to local advocacy efforts, social media allows practices to be more personable to prospective patients.
Raise awareness on public health topics
Most major and minor events come with various health risks to consider. Even something as common as the start of summer presents new dangers for vulnerable communities.

Providers are responsible for boosting timely, factual information for patients needing guidance. That includes:
- Recommendations for those experiencing symptoms or side effects
- Self-help and external resources to navigate a diagnosis
- Links to new studies and treatment options
Healthcare and social media go hand-in-hand when it comes to raising awareness about public health topics. A standard health advisory can be repurposed into countless types of content, increasing the likelihood of it resonating with its target audience.
Mitigate panic during crises or emergencies
The internet takes to a health crisis like a spark takes to a ball of lint. It doesn’t take long for feeds to spiral into misinformation and panic.
Healthcare providers are responsible for guiding the public during crises. They act as a broadcast network for vetted updates people can trust.
Sparse social profiles can reduce credibility, hindering your ability to support the community. That’s why it’s crucial to have an active social media presence as part of any crisis response plan. Established social profiles provide a platform to share important information, address community concerns and even coordinate relief efforts.
Attract top talent
As providers continue to grapple with staffing shortages, attracting talent has become one of most important applications of social media for healthcare organizations.
Social networks provide a platform to shape and strengthen your employer brand. Sharing updates on awards and behind-the-scenes content can get candidates excited about working with your organization, expanding your talent pool and expediting the hiring process.
Sharpen communications on and offline
Healthcare marketers can use social listening to gain a clear understanding of their audience’s sentiments, opinions and needs.
For example, say your community is facing a recent outbreak of Hand-foot-and-mouth disease. Using a social listening tool, an organization can track conversations relevant to the outbreak. That data can then be used to identify common questions around the diagnosis, as well as any false claims on treatments gaining traction online.
These insights can be used to tailor communication across channels so that they’re rooted in exactly what your community needs.
Choosing the right social media platforms for healthcare
Healthcare organizations succeed across multiple platforms by matching content strategy to audience demographics and communication goals. Your platform selection determines your reach and engagement success.
Facebook remains the dominant platform for reaching broad, multi-generational healthcare audiences. Use it to connect with local community groups and share important health updates.
Facebook’s community reach and demographic breadth make it a powerful platform for health awareness campaigns. While Meta has limited sensitive health-related ad targeting, marketers can still use location, age and interest-based criteria to responsibly reach their intended audiences.
Instagram’s visual format works for wellness tips, patient success stories and behind-the-scenes content. The platform resonates particularly well with younger patients and helps healthcare organizations showcase their culture through compelling imagery.
LinkedIn serves as your platform for establishing thought leadership and attracting medical talent. Share research findings, industry insights and professional development content.
It’s also your strongest channel for recruiting healthcare professionals in a competitive job market.
TikTok offers a powerful channel for reaching younger audiences with accessible, educational short-form video. Healthcare organizations are using the platform to break down complex health topics, combat medical misinformation and showcase authentic perspectives from medical professionals.
The Mayo Clinic’s nursing team exemplifies how healthcare brands can bring approachability to typically clinical topics.
Focus your energy on the platforms your patients use most. A targeted presence on two or three relevant platforms delivers more impact than a scattered approach across every network.
Social media for healthcare marketing best practices
Given that the role of social media in healthcare primarily focuses on educating followers, you’re afforded the opportunity to focus on meaningful, compelling content first and foremost. Let’s look at some examples below.
1. Publish educational content to keep patients in the loop
Whether it’s blog posts, videos or general health tips, anything you can do to provide your followers with bite-sized information is a plus.
Health tips and fast facts are especially helpful when a timely topic is driving conversation relevant to your organization. For example, after NFL player Damar Hamlin mid-game collapse in January 2023 was attributed to a rare condition called Commotio cordis, football fans and casual viewers alike began to search for more information on its causes and symptoms.
The American Heart Association capitalized on this interest by creating content around the topic designed to increase awareness and emphasize the importance of cardiopulmonary resuscitation (CPR) training.

Next time you can chime into a trending health conversation with expertise and advice, do it. Your followers will appreciate it.
2. Use inspirational content to motivate followers
Healthcare social media accounts don’t have to be boring or purely educational. You have the privilege of working in an incredibly fast-paced field that makes a real, tangible difference in people’s lives.
Your social accounts should celebrate that.

Showcase patient wins, staff celebrations and any other moments that highlight the good work done by your team. These posts provide your audience with a much-needed dose of inspiration by giving them something to rally around.
3. Post infographics for increased engagement
Mastering infographic marketing is a must-do for those working in social media and healthcare. Infographics allow healthcare marketers to break down otherwise dense information in an engaging and memorable way.

Source: Facebook
Next time a report or study is making waves within your organization or specialty, consider how you might be able to present that information visually. You can even work with creative teams to build templates with your preferred design tool, so you can produce them quickly without any blockers.
4. Harness the power of health-related hashtags
Hashtags are a staple of social media at large. Healthcare companies have plenty of opportunities to use them, too.
Whether it’s your own organizational hashtags (Northwestern Medicine’s #NMBetter, for example) or healthcare-specific tags (#CancerSucks), promoting a tag does double duty of extending your content’s reach while also making it easier for followers to share that content.
Hashtag holidays are a major opportunity for healthcare providers. Virtually every focus has its own awareness or observance period, creating ample opportunities to reach a wider audience.
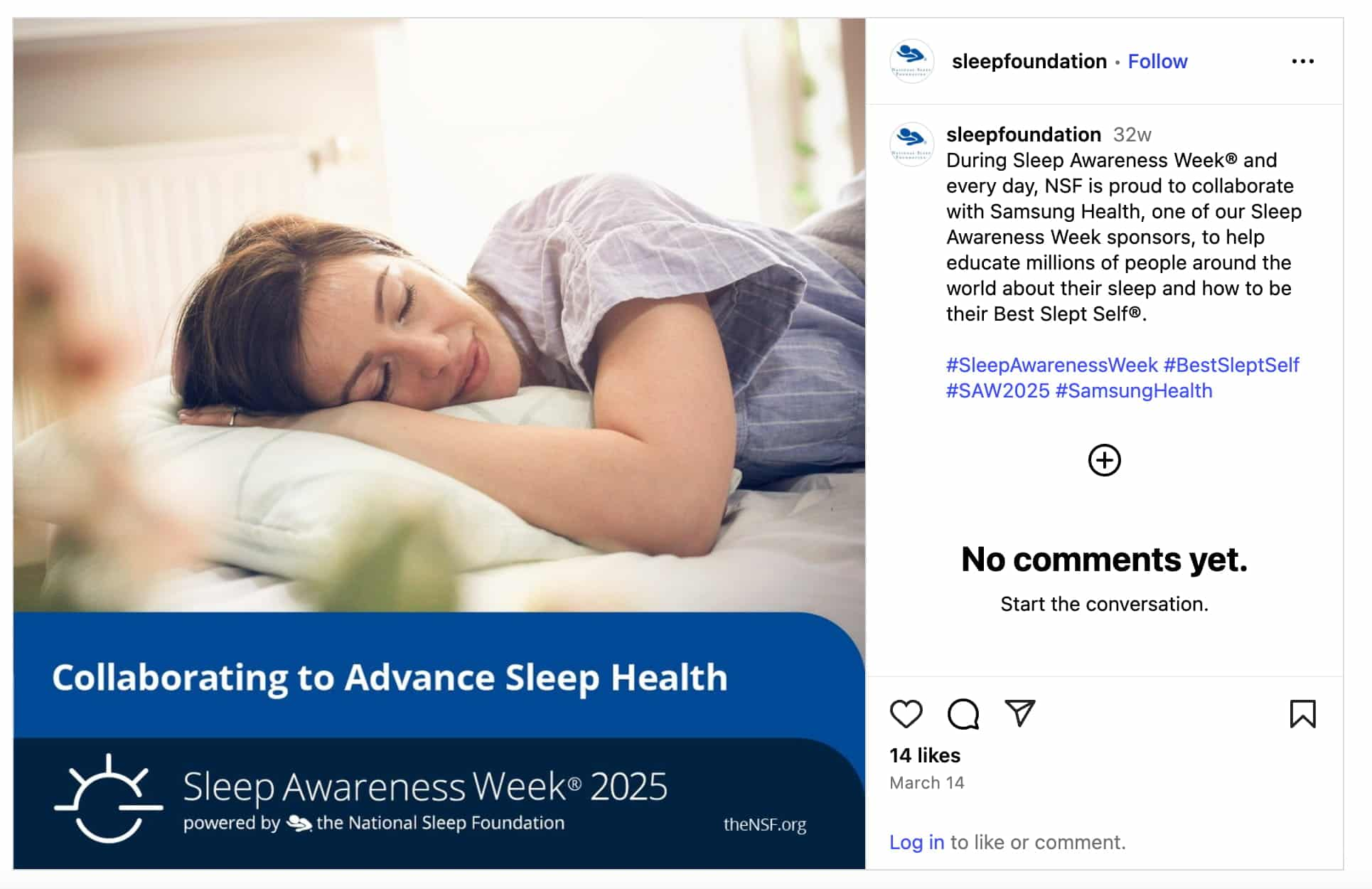
Source: Instagram
A social listening tool can help healthcare organizations zero in on the hashtags and keywords that will increase reach and make an impact with audiences. Create topics around your specific organization or trending healthcare topics within your community.
The data surfaced can help you uncover new opportunities to connect with your target audience.
5. Take your audience behind-the-scenes
Giving your various audiences—whether that be prospective patients, volunteers or staff—a peek behind the curtain can increase their interest and give your organization a more human appeal.
If you’re looking for inspiration, look to the nurses at Mayo Clinic on TikTok. They’ve found a way to bring some levity into the hospital setting through short-form videos that showcase the friendly faces that make up the organization.
These small glimpses of your practice’s day-to-day life are crucial for making a connection with followers.
6. Publish patient shout-outs to spread positivity
Sometimes the best way to highlight your practice is by letting others do the talking.
Through user-generated content, like testimonials or reviews, you can provide an unfiltered view of how you’re helping patients and supporters.

Source: X
Sharing user-generated content is also an important relationship-building tactic for healthcare organizations looking to build loyalty with their audience. A simple repost alongside a genuine message of appreciation can go a long way in the eyes of a patient.
Measuring healthcare social media success
To prove the value of your social strategy, you need to look beyond vanity metrics. The right measurements connect your social media efforts directly to patient outcomes and business objectives.
Track audience growth and engagement rates to confirm you’re reaching the right people. These metrics show whether your content resonates with your community and whether your health education efforts are landing.
Monitor website referral traffic from social platforms to understand how your content drives patients to learn more about your services. This data reveals which posts and platforms generate the most qualified interest in your organization.
For patient acquisition specifically, measure metrics that matter to your organization’s bottom line. Track appointment requests, contact form submissions and phone inquiries that originate from social media campaigns.
These conversion metrics demonstrate social’s direct impact on your pipeline.
Sprout Social’s analytics track the metrics that matter most to your organization:
- Engagement metrics: Monitor comments, shares and saves on health education content
- Community sentiment analysis: Use social listening to gauge public perception during health crises
- Conversion tracking: Measure appointment requests and website visits from social campaigns
- Competitive benchmarking: Compare your performance against other healthcare organizations
Healthcare organizations benefit from benchmarking their social media performance against industry standards. Track engagement rates, reach and conversions to identify areas for improvement and set realistic growth targets based on your organization’s baseline performance.
Report these metrics to leadership in language they understand. Connect social media performance to organizational priorities like patient satisfaction scores, community health outcomes and competitive positioning.
When you demonstrate social media’s direct impact on patient engagement and brand reputation, you secure the resources needed to expand your efforts.
HIPAA compliance and social media
To wrap things up, we need to talk about HIPAA.
The most important best practice of social media in healthcare is ensuring patient confidentiality.
In short, you need to take special care when publishing patient content which would potentially reveal sensitive information or otherwise violate HIPAA. For example, did you know that using your patients as part of your marketing materials requires explicit written consent?
There’s a lot that goes into HIPAA compliance on social media, but here are some of the basics:
- You’re allowed to engage with clients via social, but doing so means being sensitive about health information shared via direct message.
- Publishing patient stories or photos requires written consent.
- Posting sensitive or identifying information publicly can land you in trouble.
Sprout Social’s Smart Inbox centralizes patient inquiries from all platforms, allowing healthcare teams to respond quickly while maintaining HIPAA compliance. The platform’s tagging system helps categorize messages by urgency, department or health topic for streamlined workflow management.
Social media keeps your organization in good health
Healthcare providers need to meet their communities where they are: on social media.
Thoughtful use of social media in healthcare can tackle some of the industry’s biggest challenges. It’s where leading organizations are connecting with communities, building their reputations and becoming local providers (and employers) of choice.
The healthcare companies that embed social throughout their organizations will reap returns, but not without the right infrastructure. Start a free trial of Sprout Social to see how our healthcare-focused features streamline patient communication, ensure HIPAA compliance and measure real health outcomes.
FAQs about Social Media in Healthcare
Which social media platform is best for healthcare organizations?
No single platform is “best”—use Facebook for community outreach, Instagram for visual content, LinkedIn for professional networking and TikTok for younger demographics.
How do healthcare organizations measure social media ROI?
Track patient inquiries, appointment bookings, website traffic from social platforms and brand sentiment improvements to measure healthcare social media ROI.
Can healthcare providers respond to patient questions on social media?
Yes, but address general questions publicly and immediately move personal health discussions to private, HIPAA-compliant channels.
What's the difference between healthcare social media and general business social media?
Healthcare social media operates under strict regulatory compliance requirements like HIPAA, demands a focus on evidence-based information and requires building trust in a high-stakes environment where misinformation has serious consequences.
How often should healthcare organizations post on social media?
Consistency matters more than frequency—aim for three to five posts per week on your primary platforms and adjust based on engagement data and team resources.
Additional resources for Social Media for Healthcare
How to master social media for UK healthcare recruitment
Social media for healthcare: How to keep patients engaged in the digital age
Social media strategies for healthcare in the UK
How to realize the ROI of social media in healthcare
The power of social listening for healthcare organizations
Social media and mental health: how to find a healthy balance






Share